Point-of-Care Thyroid Diagnostics and
ABOUT
The Laboratory Services Committee of the American Thyroid Association® (ATA) conducted a survey of ATA® members to identify areas of member interest for education in pathology and laboratory medicine. In response to the results of the survey, the Lab Service Committee developed a series of educational materials to share with the ATA® membership. The topics below were ranked as high educational priorities amongst the membership.
INTRODUCTION
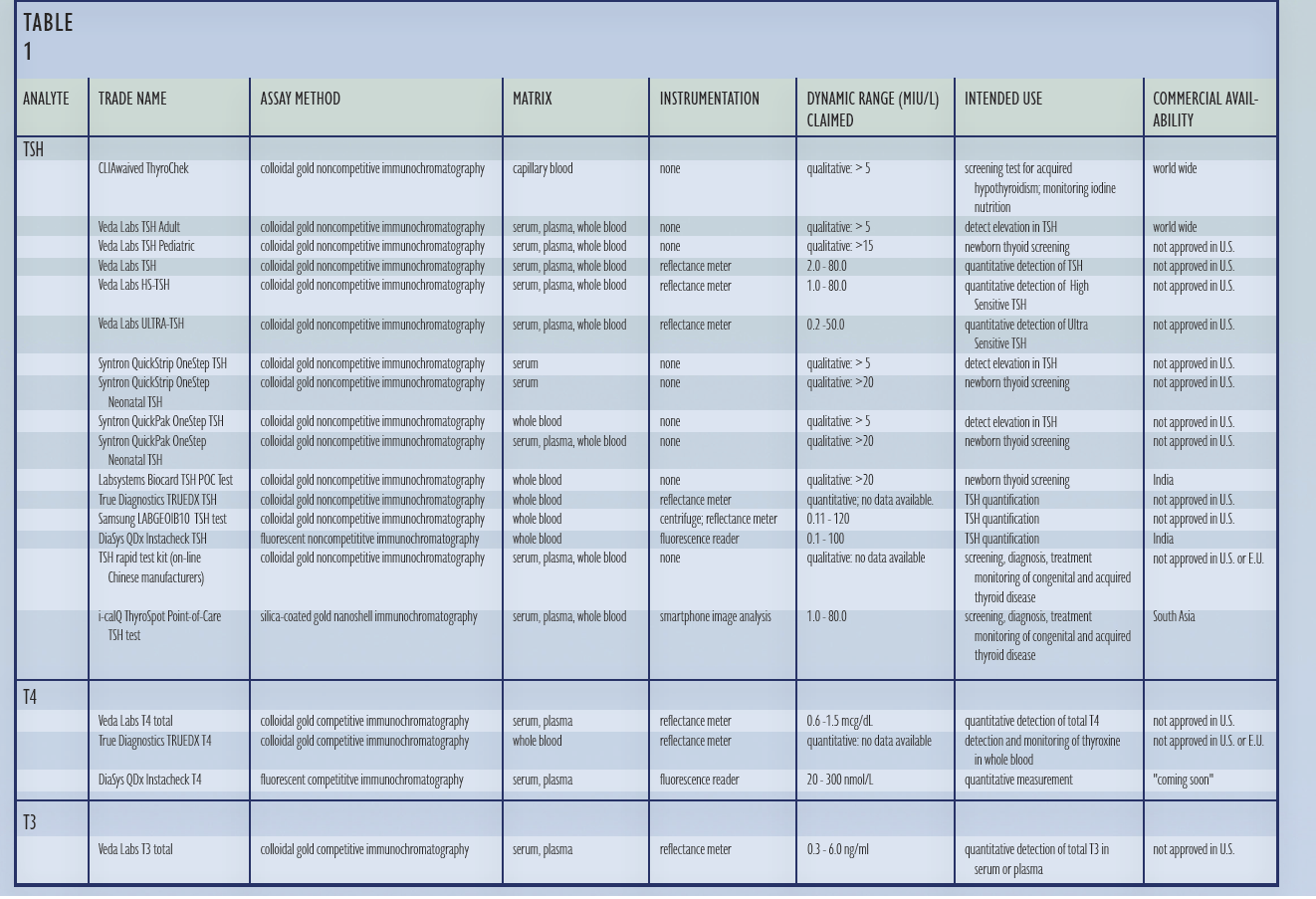
The first point-of-care thyroid diagnostic, a qualitative TSH immunochromatographic (“lateral flow”) assay, was U.S. F.D.A approved in 1999. Point-of-care quantitative TSH, T4, and T3 lateral flow assays are currently commercially available outside of the United States. This white paper summarizes the current status of point-ofcare thyroid diagnostics, provides a critical review of the technology available, and discusses trends in point-of-care thyroid diagnostics and disease management technologies. TABLE 1 lists all point-of-care thyroid diagnostic tests by analyte, measurement method, instrumentation, dynamic range, intended use, and commercial availability.
Point-of-care diagnostics are characterized by ease of use, minimal equipment requirements, rapid results, and low cost1. Point-of-care tests use less sample volume and have a quicker turnaround time compared to desktop and laboratory instrument based assays, but do not have the precision or replicability equivalent to hormone measurements performed by automated instruments. The first generation of thyroid point-of-care diagnostics, TSH qualitative lateral flow tests, employed solid phase sandwich immunoassay methods developed for measuring other glycoprotein hormones, including hCG, LH, and FSH. These tests, which are approved for sale in the US, are designed to use capillary blood to detect serum TSH concentrations above 5 mIU/L serum and use visual inspection to determine test results. Outside of the US, serum, plasma, and whole blood TSH lateral flow immunoassays are commercially available. Used with an instrument to quantify the test result, these second generation TSH assays are intended to provide a low-cost and more efficient method for measuring TSH. Based upon the commercial success of point-of-care quantitative TSH assays, rapid tests for T4 and T3 have been developed.
ASSAY PRINCIPLES
TSH is measured using a pair of antibodies that recognize distinct epitopes on the beta TSH subunit and intact TSH. TSH-specific antibody pairs provides excellent specificity and no significant crossreactivity with other glycoprotein hormones. T4 and T3 are measured using a competitive, single-antibody immunochromatographic assay format. Information on the specificity and sensitivity of commercially available T4 and T3 assays is not available. All TSH, T4, and T3 assays, including chemilumenscent, fluorescent, and immunochromatographic immunoassays, generate an optical signal that is proportional to the amount of hormone present in an aliquot of serum. By quantifying the optical signal, the amount of hormone present in an aliquot of serum is measured. Quantifying optical signals can be done with the naked eye, photoelectric cells, or CCD/CMOS chips.
AVAILABLE TESTS
Point-of-care qualitative whole blood TSH assays, designed to detect TSH >5 mIU/L and intended for use as a screening test for primary hypothyroidism in children and adults and for monitoring iodine nutrition, are currently available in the United States. Clinical laboratory regulations in the US categorize diagnostic tests which use serum/plasma instead of whole blood as too complex for use outside of a clinical laboratory environment2. As a result, serum and plasma-based quantitative point-ofcare TSH, T4, and T3 lateral flow assays are not available in the U.S., but are available in Europe, Latin America, Africa, and Asia.
NEXT GENERATION POINT-OF-CARE
TECHNOLOGIES: THYROID DISEASE MANAGEMENT
- Quantitative point-of-care TSH assays can measure TSH with precision sufficient for the diagnosis and management of primary hypothyroidism.
- Congenital hypothyroidism represents an endocrine emergency. The rapid turnaround time, minimal sample volume, and elimination of hematocrit bias make point-of-care TSH assays the method of choice for newborn thyroid screening3.
- Point-of-care TSH assay affordability and ease of use enables thyroid diagnostic testing in resource-limited settings4.
- Devices used to read point-of-care TSH tests incorporate computational and communications electronics to provide Artificial Intelligence rules-based patient-specific test interpretation and decision support and integrate with laboratory information systems.
CAVEATS
- Naked eye reading of point-of-care tests is subjective and unreliable, as the optical signal detection is affected by ambient light, position of the test, and visual acuity.
- Instrument-read point-of-care tests control illumination, test position, and light detection to provide more precise and reproducible results5.
- Because of hematocrit variability, point-of-care TSH tests which use serum / plasma are more accurate than tests which measure TSH in a capillary or whole blood sample6.
- Independent verification of test performance is not available for non- U.S. F.D.A. approved tests, thus substantiation of the performance of point-of-care quantitative T4 and T3 assays is not available.
- Independently verify test performance in the intended use setting; do not believe manufacturer’s claims.
- A poorly performing point-of-care test read by an instrument is still a poorly performing point-of-care test.
SUMMARY AND RECOMMENDATIONS
- Serum or plasma quantitative TSH assays are more accurate, affordable, accessible, and clinically useful than dried blood spot TSH measurement for newborn thyroid screening.
- Whole blood qualitative TSH tests are unreliable for detecting TSH < 10 mIU/L and inaccurate if the blood sample has a hematocrit > 50%.
- Point-of-care TSH tests that use a known starting volume of serum or plasma have a precision equivalent to lab instrument TSH tests.
- Currently available qualitative point-of-care TSH tests can detect TSH > 10 mIU/L in a whole blood sample with a hematocrit < 50%. These tests are useful to screen for primary hypothyroidism7.
- Quantitative point-of-care TSH tests which use a defined starting volume of serum / plasma claim to measure TSH in the range 0.1 – 100.0 mIU/L. Independent verification of test performance by the end user is required.
- Point-of-care thyroid diagnostic products have a role in thyroid disease screening, diagnosis, and management8.
REFERENCES
- Kost GJ. Goals, guidelines, and principles for point-of-care testing. In: Kost JG, editor. Principles and practice of point-of-care testing. Philadelphia: Lippincott Williams and Wilkins; 2002. p. 3–12.
- Kramer DB, Xu S, Kesselheim AS. Regulation of medical devices in the United States and European Union. N Engl J Med 2012;366:848–55.
- Ehrenkranz J, Kappy M, Levine M. Validation of a Smartphone TSH Immunoassay for Point-of-Care Newborn Thyroid Screening. Int. J. Neonatal Screen. 2016, 2, 5
- Fualal J, Ehrenkranz J. Access, availability, and infrastructure deficiency: The current management of thyroid disease in the developing world. Rev Endocr Metab Disord. DOI 10.1007/s11154-016-9376-x
- You D, Park T, Yoon J-Y. Cell-phone-based measurement of TSH using Mie scatter optimized lateral flow assays. Biosensors and Bioelectronics 40 (2013) 180–185
- Hall E, Flores S, De Jesu´s V. 2015 Influence of hematocrit and total spot volume on performance characteristics of dried blood spots for newborn screening. Int J Neonatal Screen 1:69.
- Puddermann A, Horizon Scan Report 0029 Date: 23 May 2013 Diagnostic Technology: Point-of-care testing for thyroid stimulating hormone 2013; Horizon Scan Report 0029. https://www.oxford.dec.nihr.ac.uk/files/reports…/ horizon-scanning-report0029-tsh.pdf. Last accessed 16 Feb. 2018.
- Ehrenkranz J. 2017 Point-of-Care Endocrine Diagnostics. Endocrinol Metab Clin N Am 46; 615–630.
FURTHER INFORMATION
For more information on the survey findings, see Thyroid, Vol. 27, No. 12. https://www.thyroid.org/professionals/ata-publications/