THYROID CANCER TUMOR BOARD
Discordant Cytopathological Diagnosis of Well-Differentiated Thyroid Cancer Affects Patient Treatment
Wendy Sacks
Clinical Thyroidology
CASE SUMMARY • • • • • • • • • • • • • • • • • • • • • • • •
A teenage girl had been diagnosed with Hashimoto’s thyroiditis and a thyroid nodule about 2 years ago. She has a family history of autoimmune thyroid disease, and a grandmother also had a goiter. Her thyroid function was normal on presentation. A neck ultrasound revealed a 3-cm nodule Fine-needle aspiration under ultrasound guidance was performed and cytology was consistent with follicular lesion of undetermined significance (FLUS). She sought several opinions for management of this nodule, and ultimately a decision was made to monitor it. Nine months later, a repeat ultrasound demonstrated growth of the nodule that then measured 4 cm in maximum dimension (Figure 1). A repeat biopsy with molecular testing revealed follicular neoplasm, benign Afirma gene expression classifier and negative MiRInform molecular panel. Because of the increase in nodule size and indeterminate cytology, a hemithyroidectomy was performed. Surgical pathology demonstrated a 4.5-cm minimally invasive follicular thyroid carcinoma (FTC) with a focus of vascular invasion. The margins were free of tumor. Also noted were foci of cellular architectural atypia, including insular and/or solid patterns.
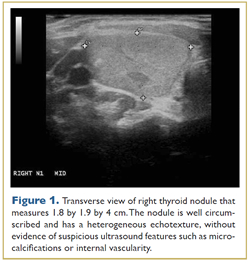 |
| Larger Image |
A young patient with minimally invasive FTC typically has an excellent prognosis, but the focus of vascular invasion and insular components suggest a more aggressive tumor. The family requested a second review of the pathology, which was sent to an international expert. The second reviewer agreed with the assessment, reporting “We are dealing with a carcinoma of follicular cells which exhibits both capsular and vascular invasion. The latter is rather impressive in the sense of involving a large vessel located well outside the capsule of the tumor and attached to the wall of the vessel. There is also evidence of capsular invasion in adjacent areas.” He continues, “the other important point relates to a focal insular pattern of growth, focal mitotic activity, and necrosis. I believe that these features are those of a poorly differentiated carcinoma focally developed within a better-differentiated lesion. My diagnosis therefore is follicular carcinoma with capsular and vascular invasion and poorly differentiated foci. This tumor is likely to follow an aggressive clinical course.”
The patient underwent a completion thyroidectomy; there was no thyroid cancer in the contralateral lobe. Her family sought multiple opinions as to whether she should have postoperative treatment with radioactive iodine (RAI). Recommendations ranged from treating with 100 mCi to treating with 50 mCi to no RAI treatment (hormone suppression alone). After much discussion about the risks and benefits of 131I, the family opted for the 100-mCi dose. Just before the treatment, the patient sought another opinion; during slide review, the pathological diagnosis changed to encapsulated FVPTC [follicular variant papillary thyroid carcinoma] without capsular involvement.
ANALYSIS AND COMMENTARY • • • • • •
The discrepancy between opinions in the interpretation and diagnosis of surgical pathology for thyroid cancer among intrainstitutional reviewers and interinstitutional reviewers has been well documented in the literature, with rates of discordance of up to 83%. Notably, this can have a significant impact on the treatment of the patient (1, 2). In one retrospective review of 66 thyroid cancer cases by pathologists at the Institute of Pathology at Leeds, there was an 18% discrepancy in the diagnosis of 66 thyroid cases diagnosed between January 2001 and March 2003 (1). The changed diagnoses altered treatment in 5 of 12 patients and altered prognosis in all 12.
An area of particular difficulty is the diagnosis of follicular thyroid adenoma (FA), follicular thyroid carcinoma (FTC) and the follicular variant of PTC (FV-PTC) (1, 3). FTCs are divided into two major categories based on the degree of invasiveness: minimally invasive FTC and widely invasive FTC. The differential diagnosis of minimally invasive FTC includes FV-PTC, the most common subset of papillary carcinoma, found in up to 56% of patients, depending on the geographic area (4, 5). FV-PTC is almost exclusively arranged in follicles lined by cells with characteristic papillary carcinoma nuclei. Furthermore, there are two categories of FV-PTC, encapsulated and invasive. Nuclear features encountered in follicular nodules often show some, but not all, of the nuclear features of PTC, making a clear-cut diagnosis of minimally invasive FTC or FV-PTC difficult. (3). A Mayo Clinic study in which 10 expert pathologists reviewed 87 cases initially diagnosed as FV-PTC (n = 84), FA (n = 2), and FTC (n = 1) demonstrated observer variability. There was 39% concordance from 10 expert pathologists after review of the same slides; concordance improved to 66.7% for cases with metastatic disease (6).
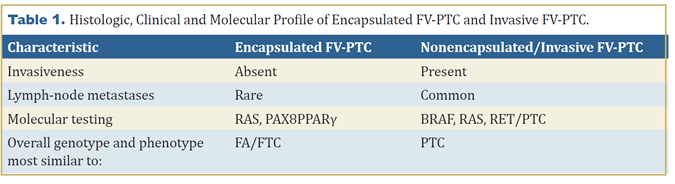
In addition to a similar invasive pattern (or lack thereof), encapsulated FV-PTC and minimally invasive FTC have other similarities. They rarely metastasize to lymph nodes (only 5% of cases), and the molecular profile is similar (Table 1). Encapsulated FV is close to the follicular carcinoma group of tumors, with a high prevalence of RAS mutation and very low BRAF mutation rate. Of 47 cases (28 encapsulated FV and 19 infiltrative FV) retrospectively reviewed at Memorial Sloan-Kettering from 1980 to 2002, a total of 11 of the 28 encapsulated FV had a mutation (10 RAS and 1 PAX8PPARγ) and 9 of 19 of the infiltrative FV group harbored a mutation (5 BRAF, 2 RAS, 2 RET/PTC) (7, 8). While molecular genotyping can better classify these tumors into clinically relevant entities, morphologic examination and interpretation remains the final determinant of categorization in a majority of cases.
Liu et al. at Memorial Sloan-Kettering reviewed prognoses for FV-PTC in a series of 78 patients between 1980 and 1995 (9). Sixty-one patients had encapsulated FV and 17 had nonencapsulated FV. Patients with the encapsulated FV-PTC had the following characteristics: 98% negative margins, 5% lymph-node involvement, 5% extrathyroidal extension, 16% lymphovascular invasion, 14% capsular invasion, and no distant metastases. Over a median follow-up of 11 years, none of the 42 patients with noninvasive, encapsulated FV-PTC had recurrences or metastases or died of disease. These authors suggest that noninvasive, encapsulated FV-PTC should denote a benign clinical behavior and propose that patients with this variant may be treated with lobectomy (9).
While unanimity among diagnostic consultants is ideal, patients should know that a difference of opinion is quite common among experts. It does not mean that a mistake has been made, but undertreatment and overtreatment can occur, depending on the diagnosis.
Conclusions
The young patient described above had a growing mass with an indeterminate preoperative diagnosis and negative molecular panel. Postoperative surgical pathologic review by the local institution and an outside expert reviewer first suggested a potentially aggressive 4.5-cm follicular thyroid carcinoma with vascular and capsular invasion. A third review at a different institution revealed encapsulated, noninvasive FV–PTC, a diagnosis which has a benign clinical course. Many reports in the literature identify interobserver variability in pathologic interpretation particularly for follicular carcinomas and follicular variants of papillary thyroid cancer. Changes in clinical management occur in a significant percentage of cases depending on the pathology. Perhaps Next-Gen Sequencing (see Hershman review) will enable further diagnostic certainty preoperatively to assist in guiding management for these cases. Rather than the 100-mCi dose of 131I recommended for FTC, our patient received 30 mCi of 131I at the outside institution for FV-PTC. The posttreatment scan showed iodine uptake in the thyroid bed only, consistent with residual thyroid tissue.
References
- Hamady ZZ, Mather N, Lansdown MR, Davidson L, Maclennan KA. Surgical pathological second opinion in thyroid malignancy: impact on patients’ management and prognosis. Eur J Surg Oncol. 2005;31:74-7.
- Hirokawa M, Carney JA, Goellner JR DeLellis RA, Heffess CS, Katoh R, Tsujimoto M, Kakudo K. Observer variation of encapsulated follicular lesions of the thyroid gland. Am J Surg Pathol 2002;26:1508-14.
- Franc B, De La Salmoniere P, Lange F, Hoang C, Louvel A, De Roquancourt A, Vilde F, Hejblum G, Chevret S, Chastang C. Interobserver and intra-observer reproducibility in the histopathology of follicular thyroid carcinoma. Hum Pathol 2003;34:1092-100.
- Grogan RH, Kaplan SP, Cao H, Weiss RE, DeGroot LJ, Simon CA, Embia OM, Angelos P, Kaplan EL, Schechter RB. A study of recurrence and death from papillary thyroid cancer with 27 years of median follow-up. Surgery 2013;154:1436-47. Epub September 26, 2013.
- Passler C, Scheuba C, Prager G, Kaczirek K, Kaserer K, Zettinig G, Niederle B. Prognostic factors of papillary and follicular thyroid cancer: differences in an iodine-replete endemic goiter region. Endocr Relat Cancer 2004;11:131-9.
- Lloyd RV, Erickson LA, Casey MB, Lam KY, Lohse CM, Asa SL, Chan JK, DeLellis RA, Harach HR, Kakudo K, et al. Observer variation in the diagnosis of follicular variant of papillary thyroid carcinoma. Am J Surg Pathol 2004;28:1336-40.
- Rivera M, Ricarte-Filho J, Knauf J, Shaha A, Tuttle M, Fagin JA, Ghossein RA. Molecular genotyping of papillary thyroidcarcinoma follicular variant according to its histological subtypes (encapsulated vs infiltrative) reveals distinct BRAF and RAS mutation patterns. Mod Pathol 2010;23:1191-200. Epub June 4, 2010.
- Nikiforova MN, Biddinger PW, Caudill CM, et al. PAX8-PPARgamma rearrangement in thyroid tumors: RT-PCR and immunohistochemical analyses. Am J Surg Pathol 2002;26:1016-23.
- Liu J, Singh B, Tallini G, Carlson DL, Katabi N, Shaha A, Tuttle RM, Ghossein RA. Follicular variant of papillary thyroid carcinoma: a clinicopathologic study of a problematic entity. Cancer 2006;107:1255-64.
CLINICAL THYROIDOLOGY • DECEMBER 2013 VOLUME 25 • ISSUE 12 • © 2013



